The Wound Care Gel Recall 2026 has been officially announced by the U.S. Food and Drug Administration after Blaine Labs, Inc. issued a voluntary nationwide recall of specific wound care gel products.
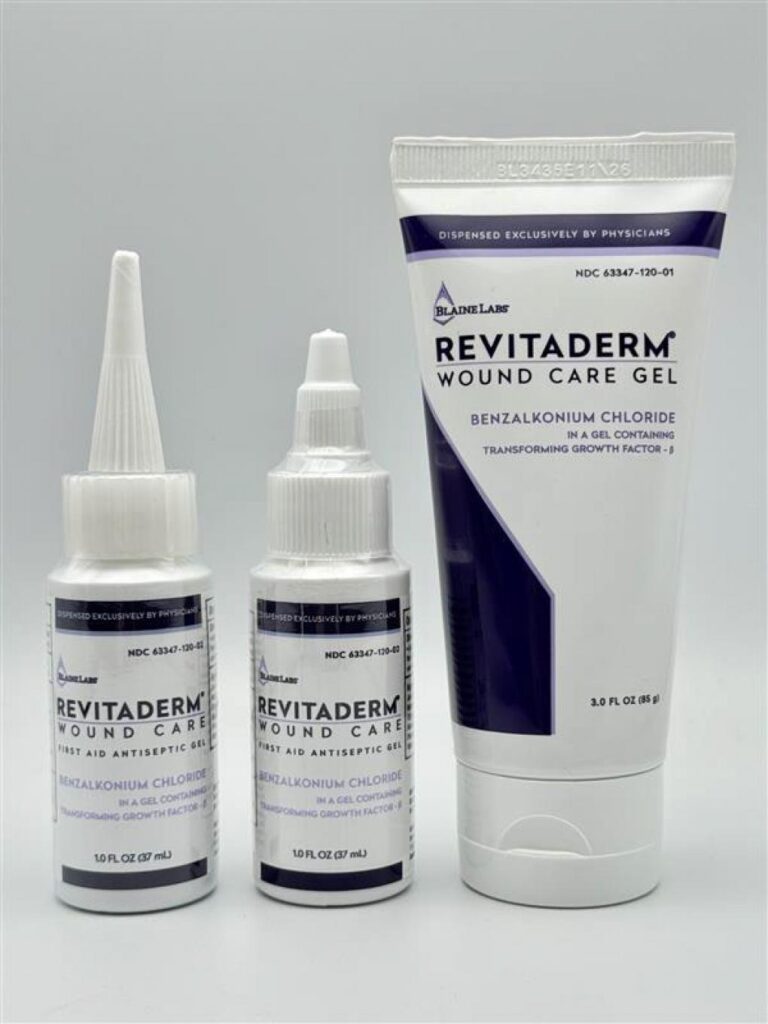
This Wound Care Gel Recall 2026 involves Revitaderm and Tridergel antiseptic gels that were found to be contaminated with bacteria, making them unsafe for use on open skin or wounds.
Affected Products in the Wound Care Gel Recall 2026
Consumers and healthcare providers should carefully check for the following products included in the Wound Care Gel Recall 2026.
Revitaderm® Wound Care Antiseptic Gel
Active ingredient: Benzalkonium Chloride 0.1%
Revitaderm products can be identified by purple trim and are available in both bottles and tubes.
1 oz Bottle (Two Cap Styles)
- Short twist cap
- Long pointy cap
- Lot number and expiration date printed on the bottom
Affected lots:
- BL3608 — Exp. 07/01/28 — Yellow ink — Short twist cap
- BL3435 — Exp. 11/06/26 — Black ink — Long pointy cap
- BL3525 — Exp. 08/07/27 — Black ink — Long pointy cap
3 oz Tube
- Lot and expiration date printed on the top crimp
Affected lots:
- BL3435 — Exp. 11/06/26
- BL3525 — Exp. 08/07/27
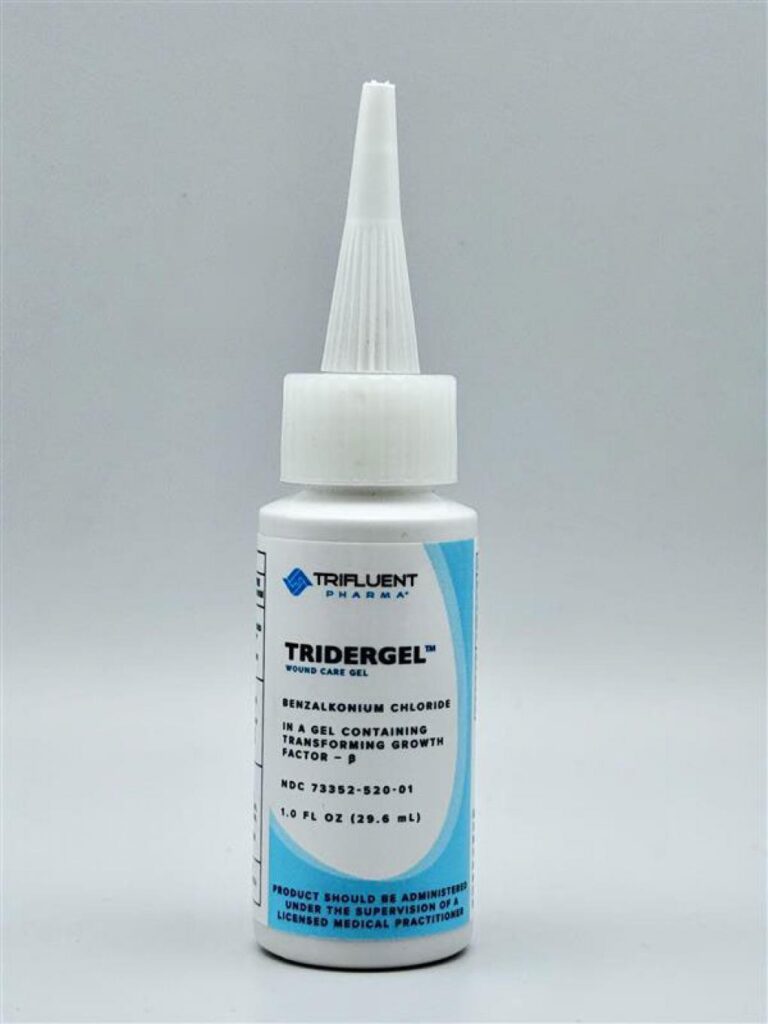
Tridergel™ Wound Care
Active ingredient: Benzalkonium Chloride 0.1%
Tridergel products can be identified by light blue trim.
1 oz Bottle
- Long pointy cap
- Lot number printed on bottom
Affected lot:
- BL3435 — Exp. 11/06/26
Why the Wound Care Gel Recall 2026 Matters
The Wound Care Gel Recall 2026 was issued after testing found contamination with Lysinibacillus fusiformis, a type of environmental bacteria.
This contamination can:
- Enter the body through open wounds
- Cause infections
- Delay healing
- Lead to complications in vulnerable individuals
The Wound Care Gel Recall 2026 is especially important for:
- Elderly individuals
- Diabetic patients
- People with weakened immune systems
- Anyone with open or chronic wounds
Understanding the Risk Behind the Wound Care Gel Recall 2026
The bacteria identified in the Wound Care Gel Recall 2026 may not be visible to the eye, meaning the product can look normal but still be unsafe.
In some cases, exposure could lead to:
- Skin irritation
- Infection
- Serious complications such as bloodstream or systemic infections
This is why the Wound Care Gel Recall 2026 requires immediate action even if no symptoms are present.
What You Should Do Right Now
If you have any products included in the Wound Care Gel Recall 2026, take the following steps:
✔ Stop Using the Product Immediately
Do not apply the gel to any wound or skin condition.
✔ Check the Lot Number Carefully
Look at:
- Bottom of bottles
- Top crimp of tubes
✔ Contact Blaine Labs
- Phone: (800) 307-8818
- Email: Contact@blainelabs.com
✔ Return or Dispose of the Product
Follow the company’s instructions for safe handling.
Important Distribution Detail
One key detail in the Wound Care Gel Recall 2026 is that these products were distributed to healthcare providers and not sold directly in retail stores or online.
This means:
- You may have received the product from a doctor’s office
- It may not be something you purchased yourself
What People Are Asking About the Wound Care Gel Recall 2026
Were these sold in stores?
No. The products in the Wound Care Gel Recall 2026 were distributed through healthcare providers.
What if I already used the product?
Monitor for symptoms such as redness, swelling, or irritation. Seek medical care if needed.
Where do I find the lot number?
- Bottles: bottom
- Tubes: top crimp
Keeping Each Other Informed
Have you ever received medical products directly from a healthcare provider and not thought twice about checking them?
The Wound Care Gel Recall 2026 is a reminder to always double-check—even for products you didn’t buy yourself.
Stay updated with the latest recalls in our Consumer Safety Alerts section. https://everydayanswers.online/wp-admin/post.php?post=1197&action=edit
For full official details, visit the FDA recall notice. https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/blaine-labs-inc-issues-voluntary-nationwide-recall-wound-care-gel-products-due-microbial
Final Thoughts
The Wound Care Gel Recall 2026 highlights the importance of paying attention to product recalls, especially for items used on the body.
Even everyday medical products can pose risks if contamination occurs. Taking a few minutes to check your supplies can make a significant difference in protecting your health.
