A TecFlox Serum Recall 2026 has been issued due to a risk of child poisoning associated with non-child-resistant packaging. The main reason for the TecFlox Serum Recall 2026 is that the packaging does not meet child-resistant safety requirements.
The recall involves hair and beard growth serum bottles that are not child-resistant, which violates federal safety standards and could lead to serious injury or death if swallowed by young children.
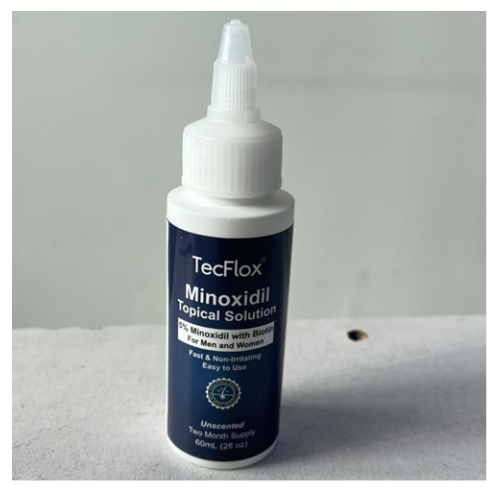
Consumers who own this product should immediately secure the bottles out of reach of children and follow the recall instructions to receive replacement bottles.
Why the TecFlox Serum Recall 2026 Was Issued
The hair and beard growth serum contains minoxidil, which is a medication commonly used for hair growth treatments. Products containing minoxidil are required by law to be sold in child-resistant packaging under the Poison Prevention Packaging Act.
The recalled bottles are not child-resistant, which means children could easily open the bottles and ingest the contents, creating a poisoning risk that could result in serious injury or death.
This is why the recall was issued, even though no injuries have been reported at this time.
TecFlox Hair and Beard Growth Serum Recall 2026 Details
This recall involves:
- TecFlox Hair and Beard Growth Serum bottles
- White plastic bottles with a white nozzle
- Sold in packs of four bottles
- Labeled “TecFlox Minoxidil Topical Solution”
- Manufacture date MFD 2025.8.25 printed on the underside of the bottle
Approximately 900 bottles are included in this recall.
The products were sold online, including on Amazon.
What Consumers Should Do About the TecFlox Hair and Beard Growth Serum Recall 2026
If you own the recalled serum bottles, follow these steps:
- Secure the bottles out of sight and reach of children immediately.
- Do not allow children to handle the bottles.
- Dispose of the product contents safely.
- Take a photo of the bottle in the trash showing the manufacture date.
- Email the photo to the company to receive replacement bottles.
Consumers should contact the company at:
TecFloxrecall@outlook.com to receive free replacement bottles.
Only bottles with product remaining will be replaced.
Why Child-Resistant Packaging Is Required
Child-resistant packaging is required for certain products that contain chemicals, medications, or substances that could be harmful if swallowed by children.
Minoxidil is a medication that can cause serious health problems if ingested by children, which is why federal law requires special packaging that children cannot easily open.
When companies fail to use child-resistant packaging, products can be recalled because the packaging itself creates a safety hazard.
Important Safety Reminder for Parents
This recall is a reminder that many household products, cosmetics, and medications can be dangerous to children if swallowed.
Parents and caregivers should:
- Keep medications and topical treatments out of reach
- Store products in locked cabinets when possible
- Never leave bottles on counters or nightstands
- Check product packaging for child-resistant caps
- Pay attention to product recalls involving household items
Many recalls involving poisoning hazards are related to packaging rather than the product itself.
Related Recalls Involving Similar Products
There have been several recalls in recent years involving hair growth serums and other products containing minoxidil that were not packaged in child-resistant bottles. These recalls show that packaging safety requirements are taken very seriously because children can accidentally ingest these products.
This is why consumers should always check recall notices, especially for household products, medications, and cosmetics.
TecFlox Serum Recall 2026 Safety Reminder
Product recalls are issued to prevent injuries and protect consumers. Many recalled products remain in homes because people do not realize their product has been recalled.
It is always a good idea to:
- Check recall websites regularly
- Register products when possible
- Stop using recalled products immediately
- Share recall information with friends and family
What Consumers Should Do About the TecFlox Serum Recall 2026
Many people store hair products, medications, and cosmetics in bathrooms or bedrooms where children may have access to them.
Do you store medications and topical products in child-safe areas at home? Most people don’t realize how dangerous some everyday products can be until a recall is issued.
Sharing recall information can help prevent accidents and injuries.
According to the U.S. Consumer Product Safety Commission, the bottles are not child-resistant and could allow children to access the product, creating a poisoning risk. https://www.cpsc.gov/Recalls/2026/TecFlox-Hair-and-Beard-Growth-Serum-Bottles-Recalled-Due-to-Risk-of-Serious-Injury-or-Death-from-Child-Poisoning-Violates-Mandatory-Standard-for-Child-Resistant-Packaging
Recent recalls have involved electronics, furniture, clothing, and household products, which shows why consumers should regularly check product recall and safety alerts. https://everydayanswers.online/?p=1172
The TecFlox Serum Recall is an important reminder that packaging safety matters just as much as the product itself. The TecFlox Serum Recall involves bottles that do not meet child-resistant packaging requirements, which can put children at risk if the product is left within reach. Anyone who has the recalled product at home should act quickly and follow the company’s replacement instructions.
Final Thoughts
The TecFlox Hair and Beard Growth Serum Recall 2026 was issued because the bottles are not child-resistant, creating a serious poisoning risk for children. If you own this product, secure it immediately and follow recall instructions to receive replacement bottles.
Product recalls are issued to prevent injuries and protect families, and staying informed about recalls is one of the easiest ways to keep your home safe.
